Medexprim is now a proud member of BC Platforms.
Follow us on:
We help accelerate precision medicine
and find the right treatment
for the right patient
at the right time
Through multicentric curated datasets of imaging
and associated clinical data.
We want to make Europe a major player in precision medicine,
in oncology, cardiology, neurology, and rare diseases.
We unlock the
power of data
to meet clinical research challenges and
guarantee interoperability of health data
guarantee interoperability of health data
We build bridges between
Hospitals and Biopharma
to pioneer new ways to design and
optimize your clinical trials
The right treatment
for the right patient
at the right time
Through the Medexprim software Suite™ deployed on premise, linked with our cloud platform R-WiDE®.
European
by design
We promote CIAN studies:
Cohort Imaging Analytics Networks
Medexprim helps develop prognostic and predictive models with Real-World cohorts.

We co-lead and contribute to multicentric European consortia
Medexprim participates in three EU-funded projects:


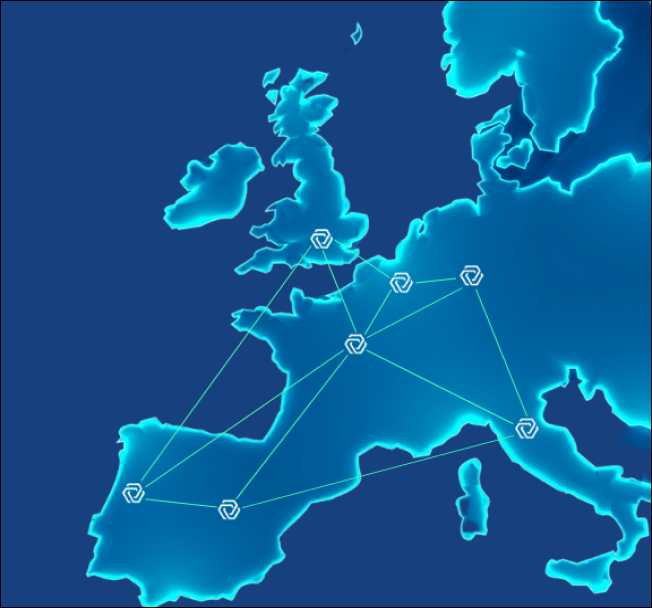
We federate a network of
Hospitals, Cancer Centers, and KOLs
Hospitals, Cancer Centers, and KOLs
Medexprim fosters a European research
community to improve patient care.
community to improve patient care.

We generate exciting
Scientific insights
Scientific insights
Medexprim’s researchers, engineers, and experts are immersed daily in the
world of Life sciences and innovation. Get informed and join the conversation!
world of Life sciences and innovation. Get informed and join the conversation!
Join a
transverse
avant-garde organisation
transverse
avant-garde organisation
A shared vision and governance
4 values
Care
Ambition
Cooperation
Innovation
63%
of women
8
Nationalities
08 December 2023
BC Platforms acquires Medexprim, paving the way for Global Leadership in Real-World Data and Real-World Evidence Analytics
BC Platforms, a global leader in healthcare data management and analytics, today announced it has acquired Medexprim,...
27 November 2023
RSNA 2023: Medexprim brings European multi-imaging datasets to Innovation.
Medexprim, a leading European innovator in the field of Real-World Data and healthcare solutions, is...
23 November 2023
Medexprim appoints Doctor Mercedes Serra as Head of Data Science for Imaging
Medexprim is proud to announce the appointment of Mercedes Serra as the Head of Data Science for Imaging.
With...
20 November 2023
IQVIA clôture l’édition 2023 de son Accelerator Program, en collaboration avec des start-ups de santé européennes
IQVIA, spécialiste mondial d’analyses avancées, de solutions technologiques et de services de recherche...
10 November 2023
RSNA 2023: Medexprim brings European multi-imaging datasets to Innovation
Medexprim is set to showcase its cutting-edge, regulatory-grade multi-imaging and multimodal datasets...
17 October 2023
ESMO Congress 2023, D-1 / Medexprim spearheads Real-World Data to advance Clinical Oncology Research
Medexprim, a leading innovator in the field of Real-World Data and healthcare solutions, is pleased to...
05 October 2023
Montaine Marteau appointed as Chief Product and Technical Officer at Medexprim
Medexprim is pleased to announce the appointment of Montaine Marteau as Chief Product and Technical Officer...
03 October 2023
Revolutionizing Cancer Research: EUCAIM unveils first release of groundbreaking platform to fuel collaborative innovation and data sharing across Europe
The EUCAIM consortium and the European Commission are excited to announce the first public release of...
11 September 2023
3 questions to Coralie Carron
As a Ph.D. holder in Cellular and Molecular Biology, Coralie Carron assumes the pivotal role of Product...
18 July 2023
CIAN: Cohort Imaging Analytics Network
Medexprim enables the aggregation, curation and enrichment of multicentric and multimodal cohorts of...


